Single-molecule mass spectrometry
Posted in Mass Spectrometry on May 16, 2007
John J. Kasianowicz from NIST and his colleagues created a 2D method for mass spectrometry capable of a single-molecule analysis based on the interaction between a nanometer-scale pore and analytes. The method is a “nano” version of Wallace Coulter patented technique that detects particles suspended in solution by driving them through a capillary which is widely used to count and size red blood cells and platelets.
In the experiment the researches used different in size PEGs (polyethylene glycol molecules) that were pushed through the ~10nm channels of the planar lipid bilayer membranes by applying electric potential of 40 mV that created ionic current. MALDI-TOF mass spectra of the PEG samples were obtained in the reflectron mode with desorption/ionization produced by irradiation with pulsed UV light (337 nm) from a nitrogen laser.
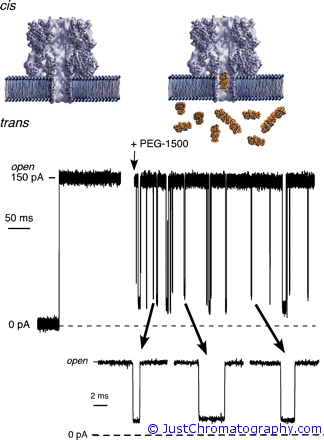
The scientist showed that a single nanopore can discriminates between polymers with different molecular masses. When a neutral PEG partitioned into a nanopore channel in a lipid bilayer membrane, it caused well defined reductions in the ionic current due to a partial-current blockades caused by the polymer in the pore. Each decrease and subsequent increase in the ionic current corresponded to a single PEG molecule entering and exiting the pore. By assigning the mean values of ionic current histograms to each blockade event, it is possible to calibrate this method and use it as a molecular sizing device, and the comparison of PEG’s mass distributions obtained with a single nanopore with a conventional MALDI-TOF showed 1:1 correlation.
This single-molecule analysis technique could prove useful for the real-time characterization of nucleic acids, proteins, or other biopolymers.
Reference: “Single-molecule mass spectrometry in solution using a solitary nanopore” by John J. Kasianowicz and et al.
DOI: 10.1073/pnas.0611085104
Tag Cloud
-
adsorbent
benzene
china
chlorophyll
chromatographic
chromatography
column chromatography
CZE
DESI
electromagnetic induction
electrospray
ELISA
eluent
explosives
forensic
furosine
Gas Chromatography
GC/MS
History
HPLC
IC
isoflavones
lab-on-a-chip
LCMS
maldi tof
Mass Spectrometry
microfluidic
nano
Nano HPLC
nanoliquid
nanoscale
nanostream
nanotechnology
nanotubes
News
organic molecule
protein
Rickettsia
rmsf
rocky mountain spotted fever
science
soy
TCM
ticks
tsvet


